Dedicated Research Staff
NWCRC employs a highly experienced and specialized research team to ensure high-quality study execution:

- 7 Board-Certified (Principal Investigators) Physicians, including Internal medicine, Family, OBGYN, Psychiatry and Neurology specialties.
- Our PIs collaborate closely with and consult with sponsors on study design and protocol development.
- 5 Licensed Medical Sub-Investigators
- 3 Dedicated Raters (experienced in psychiatric, neurological, and cognitive assessments) Additional raters as needed
- 2 Dedicated Lab Associates (certified phlebotomists)
- 10+ Study Coordinators (experienced in multi-therapeutic trials)
- 7 Regulatory Coordinators (ensuring IRB and compliance readiness)
- 6 Dedicated Recruitment Coordinators (specializing in patient outreach and engagement)
- On-Site Unblinded Staff (for investigational product (IP) management)
- On-Site locked secure IP storage room, include controlled substance, with continuous temperature monitoring, and aseptic laminated preparation hood.
Our staff’s combined expertise allows us to efficiently manage clinical trials while ensuring compliance, patient safety, and data integrity.
Our state-of-the-art facility, highly experienced investigators, and dedicated study teams ensure efficient study execution, protocol adherence, and superior patient care. We consistently hit or exceed enrollment targets, making us a reliable and high-performing site for sponsors and CROs.
At NWCRC we diligently follow the Good Clinical Practice, FDA recommendations, study protocol and IP manual instructions to accurately prepare IP, ensuring reliability throughout the trial. Our staff meticulously reviews the investigator’s brochure and relevant documentation to gain a deep understanding of the IP properties, handling requirements, and administration guidelines. When required, our staff can compound IP according to study specifications, ensuring precise formulation and dosing. Our team employs proper aseptic techniques to minimize contamination risks and maintain the integrity of the IP. We maintain detailed records of IP preparation, including lot numbers, expiration dates, and any deviations or issues encountered during the process.
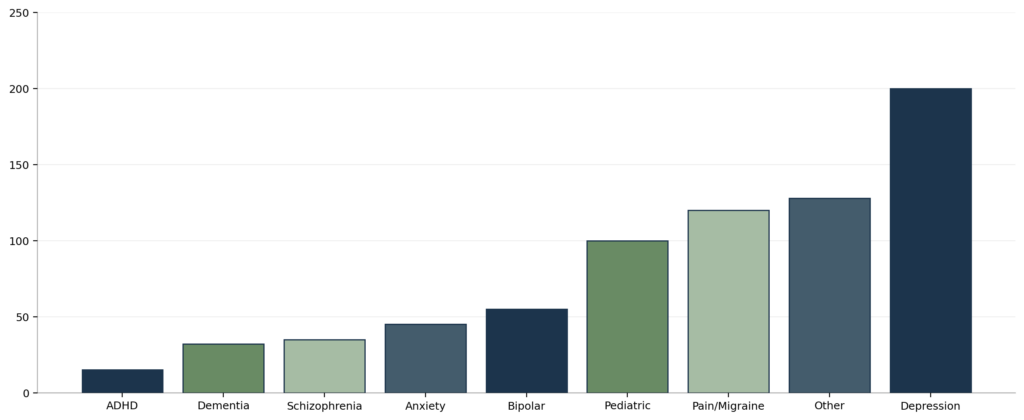
Clinical Studies by Indication
Arif Khan, MD – Medical Director & PI
Arun Arora, Wisam Al Jumaili, Gaylon Kipp, Ann Kolwitz, Aishwarya Prasad, Jeena Vaid PI’s
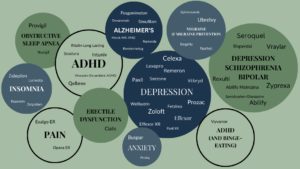
Medications NWCRC Has Worked With
We have participated extensively in the development and assessment of over 45 medications that have gone to market, providing valuable and wide-ranging benefits to the scientific and medical community.
NWCRC Sponsor Contact Form
If you are interested in partnering with us please complete the form below.
425.453.0404
1951 152nd Pl NE, Suite 200
Bellevue, WA, 98007
NWCRC Notice of Privacy Practices
NWCRC-Privacy-Notice-2021.pdf



















